LC-MS/MS Analysis of Urinary Free Cortisol and Cortisone without Matrix Interferences
Featured Application: Urinary Free Cortisol and Cortisone on Raptor Biphenyl
- Raptor Biphenyl column provides reliable separation of analytes from endogenous matrix interferences.
- Heightened selectivity for compounds that are hard to resolve on C18 and other phenyl chemistries enables accurate results.
- Fast 3-minute analysis allows high sample throughput.
Cortisol is a steroid hormone synthesized from cholesterol by a multienzyme cascade in the adrenal glands. It is the main glucocorticoid in humans and plays a critical role in glucose metabolism and immune response regulation. Cortisone, a downstream metabolite of cortisol, is measured in conjunction with cortisol to help diagnose various adrenocortical disorders, particularly hypercortisolism (Cushing syndrome). Urinary free cortisol and cortisone levels correlate well with bioactive plasma concentrations and, thus, are important biomarkers. LC-MS/MS is the preferred screening technique for these compounds because it is highly selective and helps eliminate analytical interferences that can affect immunoassay-based methods.
Due to the structural similarities between cortisol and cortisone and the potential for matrix effects from urine components, appropriate column selectivity is critical for obtaining accurate results. In the LC-MS/MS analysis of urinary free cortisol and cortisone developed here, both compounds were completely resolved from endogenous matrix interferences using a highly selective Raptor Biphenyl column. Chromatographic separation was essential in this case because some of the matrix compounds are isobaric to cortisol and cannot be distinguished by the mass spectrometer. Using a Raptor Biphenyl column and the conditions shown here, baseline separation was obtained in a 3-minute analysis, which allows labs to quickly generate more accurate quantitative data for clinical diagnosis.
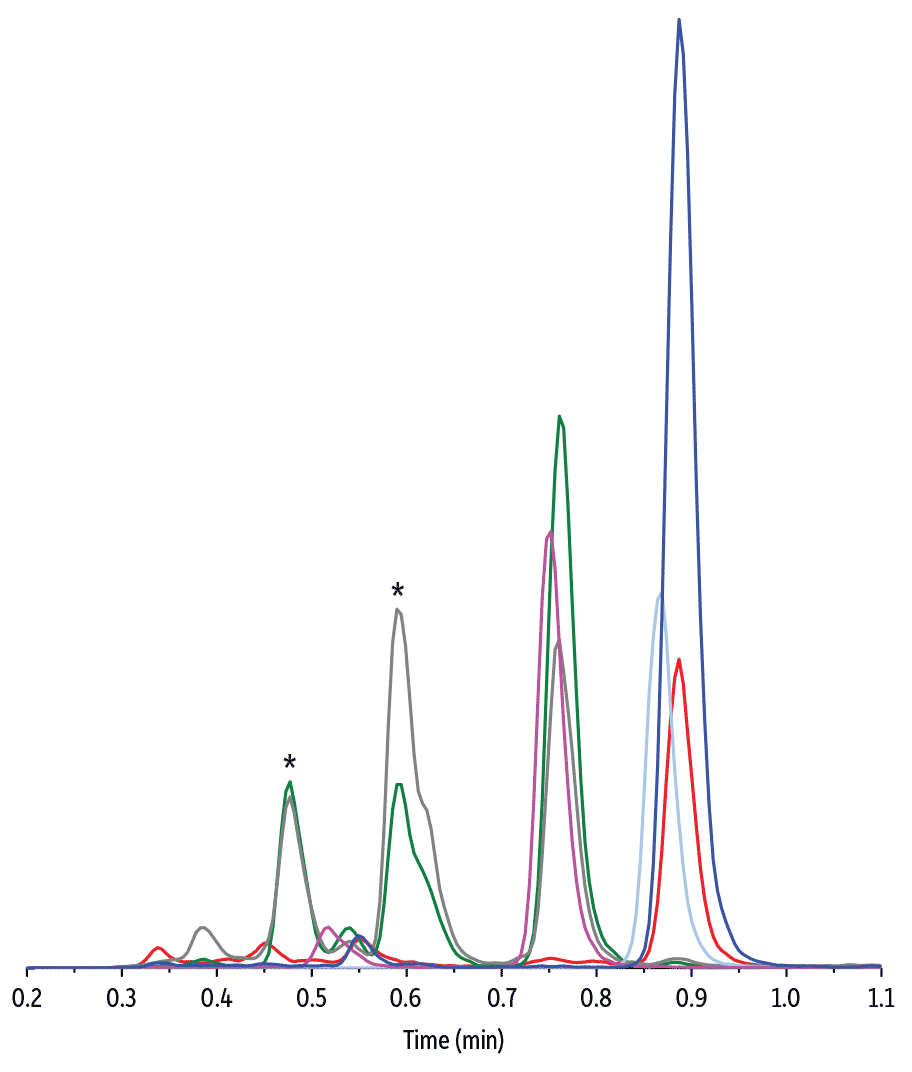
| Peaks | tR (min) | Precursor Ion | Product Ion | Product Ion | |
|---|---|---|---|---|---|
| 1. | Cortisol-d4 | 0.75 | 367.4 | 121.0 | - |
| 2. | Endogenous cortisol | 0.76 | 363.3 | 120.9 | 91.1 |
| 3. | Cortisone-d8 | 0.88 | 369.4 | 167.9 | - |
| 4. | Endogenous cortisone | 0.89 | 361.3 | 163.2 | 91.0 |
| Column | Raptor Biphenyl (cat.# 9309A52) | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Dimensions: | 50 mm x 2.1 mm ID | ||||||||||||||||||||||||||||
| Particle Size: | 2.7 µm | ||||||||||||||||||||||||||||
| Pore Size: | 90 Å | ||||||||||||||||||||||||||||
| Guard Column: | Raptor Biphenyl EXP guard column cartridge 5 mm, 2.1 mm ID, 2.7 µm (cat.# 9309A0252) | ||||||||||||||||||||||||||||
| Temp.: | 40 °C | ||||||||||||||||||||||||||||
| Standard/Sample | |||||||||||||||||||||||||||||
| Diluent: | Mobile phase A | ||||||||||||||||||||||||||||
| Conc.: | Calculated concentration is 105.1 ng/mL and 135.9 ng/mL for cortisol and cortisone, respectively | ||||||||||||||||||||||||||||
| Inj. Vol.: | 10 µL | ||||||||||||||||||||||||||||
| Mobile Phase | |||||||||||||||||||||||||||||
| A: | Water, 0.1% formic acid | ||||||||||||||||||||||||||||
| B: | Acetonitrile, 0.1% formic acid | ||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Max Pressure: | 400 bar |
| Detector | MS/MS |
|---|---|
| Ion Mode: | ESI+ |
| Mode: | MRM |
| Instrument | UHPLC |
| Sample Preparation | 1. Centrifuge female human urine for 5 min at 4500 rpm, 10 °C. 2. Aliquot 380 µL supernatant. Add 20 µL each of internal standard solution (1 µg/mL in methanol). 3. Load 200 μL of sample on to ISOLUTE SLE+ 200 µL supported liquid extraction plate (part# 820-0200-P01). 4. Apply a pulse of vacuum to initiate flow. 5. Wait 5 min for sample to completely absorb. 6. Extract samples with 1 mL of MTBE. Allow solvent to flow for 5 min under gravity. Apply vacuum for 10-30 sec to complete elution. 7. Evaporate extracts to dryness under a stream of nitrogen. 8. Reconstitute in 200 µL mobile phase A prior to analysis. 9. Vortex to mix. |

