Rapid Determination of TO-15 Volatile Organic Compounds (VOCs) in Air
Table of Contents:
Introduction
Experimental
Results and Discussion
Conclusion
Abstract
The following study evaluated the efficacy of using Nutech’s 8900DS preconcentrator in combination with an analytical system based on a 30 m x 0.32 mm x 1.00 µm Rxi-5Sil MS column for analysis of VOCs in whole air. Results were assessed based on five criteria outlined for time-integrated, whole air canister sampling in the United States (U.S.) Environmental Protection Agency’s (EPA) Compendium Method TO-15 (Determination of Volatile Organic Compounds [VOCs] in Air Collected in Specially-Prepared Canisters and Analyzed by Gas Chromatography-Mass Spectrometry [GC-MS]). Canister blank concentrations were <0.2 ppbv; the average relative standard deviation (RSD) of calibration relative response factors (RRFs) was 9.31%; average scan and selected ion monitoring (SIM) method detection limits (MDLs) were 0.06 ppbv and 35.9 pptv, respectively; average replicate precision was 4.29% difference; and average audit accuracy for all 65 targeted TO-15 VOCs was -2.82%. These performance levels for VOC analysis of air met all Method TO-15 guidelines and were achieved using a 30 m x 0.32 mm x 1.00 µm Rxi-5Sil MS column with 16.5 minute GC analysis times (~22 min total sample throughput times).
Introduction
Volatile organic compounds (VOCs) are a class of organic chemicals typically characterized as possessing relatively high vapor pressures at room temperature conditions. Their high vapor pressures result from relatively low boiling points, which cause a large number of molecules to evaporate or sublimate from liquid or solid form, respectively, into the gas phase.
VOCs are produced from both primary and secondary sources. Primary sources, either natural or anthropogenic, are directly emitted from the source to the air, while secondary sources result from atmospheric reactions. VOCs are of great interest due to their ubiquitous presence in indoor, outdoor, and personal air, and also because VOCs and their atmospheric reaction products have well known adverse environmental impacts and detrimental human health effects.
As a requirement of the Clean Air Act Amendments of 1990, the United States (U.S.) Environmental Protection Agency (EPA) is required to control 189 hazardous air pollutants (HAPs). In accordance, the EPA has generated a Compendium of Methods for the Determination of Toxic Organic (TO) Compounds in Ambient Air. More specifically, Compendium Method TO-15 (Determination of Volatile Organic Compounds [VOCs] in Air Collected in Specially-Prepared Canisters and Analyzed by Gas Chromatography/Mass Spectrometry [GC-MS]) has been developed for the sampling and analytical procedures for the measurement of a subset of 97 VOCs included in the 189 HAPs [1].
Method TO-15 is a “performance-based” guidance document for whole air canister sampling and VOC analysis; therefore, the method has very little information on specific procedures or protocols that must be followed to be method “compliant”. However, section 11.1.1 of Method TO-15 does identify three performance criteria, which should be met for a system to qualify. These criteria are: a method detection limit of ≤0.5 ppbv, replicate precision within 25 percent, and audit accuracy within 30 percent for concentrations normally expected in contaminated ambient air (0.5 to 25 ppbv).
However, there are two additional criteria laboratories should be cognizant of that are not clearly outlined in Section 11.1.1 of Method TO-15. Section 8.4.1 (Canister Cleaning and Certification) of Method TO-15 stipulates that any canister that has not tested clean (compared to direct analysis of humidified zero air of less than 0.2 ppbv of targeted VOCs) should not be used. In addition, section 10.5.5 (Technical Acceptance Criteria for the Initial Calibration) of Method TO-15 states that for the initial calibration the calculated percent relative standard deviation (%RSD) for the relative response factor (RRF) for each compound in the calibration table must be less than 30% with at most two exceptions up to a limit of 40%.
The following investigation was conducted to evaluate the efficacy of pairing Restek products with a Nutech 8900DS preconcentrator to meet the five aforementioned criteria of U.S. EPA Compendium Method TO-15. Although 60 m GC columns are typically used for TO-15 VOC analysis, a 30 m column was used here to determine if method criteria could be met with faster analysis times.
Experimental
Analytical System
For all of the experiments, the following analytical system was utilized: a Nutech 8900DS preconcentrator paired with an Agilent 6890 gas chromatograph (GC) coupled with an Agilent 5973 mass selective (MS) detector. The preconcentrator and GC-MS parameters may be found in Tables I and II, respectively.
The Nutech 8900DS preconcentrator utilizes three cryogenically cooled traps to concentrate/focus target analytes (often referred to as “micro-scale purge-and-trap”) for delivery to the GC-MS system. In order of sample flow: Trap 1 consists of glass beads used to remove water vapor, nitrogen, oxygen, and carbon dioxide (CO2) from the sample stream; Trap 2 consists of Tenax material used to focus the VOCs and remove any residual water vapor and CO2; and Trap 3 is a proprietary trap used for final analyte focusing and transfer of analytes to the GC column.
All samples were analyzed by preconcentrating 400 mL of sample with the addition of 100 mL of the TO-14A internal standard/tuning mix (cat.# 34408) (bromochloromethane, 1,4-difluorobenzene, chlorobenzene-d5, and 4-bromofluorobenzene) prepared at 20 ppbv concentrations.
Table I: Nutech 8900DS preconcentrator settings. Default preconcentration volume = 400 mL.
| Trap 1 Settings | Internal Standard | ||
|---|---|---|---|
| Cooling Temp. | -155 °C | Purge Flow | 100 mL/min |
| Preheat Temp. | 5 °C | Purge Time | 6 sec |
| Preheat Time | 0 sec | Volume | 100 mL |
| Timeout | 10 min | ISTD Flow | 100 mL/min |
| Desorb Temp. | 20 °C | ||
| Desorb Flow | 5 mL/min | Sample | |
| Desorb Time | 360 sec | Purge Flow | 100 mL/min |
| Bakeout Temp. | 200 °C | Purge Time | 6 sec |
| Flush Flow | 120 mL/min | Sample Flow | 100 mL/min |
| Flush Time | 60 sec | ||
| Sweep Flow | 120 mL/min | GC Control | |
| Sweep Time | 60 sec | Remote Start | Yes |
| Temp. Target Range | 2 °C | GC Run Time | 3 min |
| Stable Time | 60 sec | Flush Sample Line | No |
| Cooling W/He | No | GC Ready Required | Yes |
| GC Ready Timeout | 1 min | ||
| Trap 2 Settings | |||
| Cooling Temp. | -35 °C | Idle State Configuration | |
| Desorb Temp. | 190 °C | Cryotrap1 | 150 °C |
| Desorb Time | 30 sec | Transfer Line | 125 °C |
| Timeout | 10 min | Valve Oven | 125 °C |
| Bakeout Temp. | 200 °C | Cryotrap2 | 150 °C |
| Bakeout Time | 10 sec | Sampler Line | 125 °C |
| Temp. Target Range | 2 °C | Sampler Oven | 125 °C |
| Stable Time | 10 sec | Auxiliary | 125 °C |
| Cooling W/He | No | ||
| Cryofocus Settings | |||
| Cooling Temp. | -160 °C | ||
| Inject1 Time | 140 sec | ||
| Timeout | 10 min | ||
| Temp. Target Range | 3 °C | ||
| Stable Time | 10 sec | ||
Table II: Agilent 6890/5973 GC-MS parameters.
| GC Parameters | |
| Column | Rxi-5Sil MS, 30 m, 0.32 mm ID, 1.00 µm (cat.# 13654) |
| Oven | 32 °C (hold 1 min) to 150 °C at 9 °C/min to 230 °C at 33 °C/min |
| Carrier Gas | He, constant flow |
| Flow Rate | 1.5 mL/min |
| MS Parameters | |
| Transfer Line Temp. | 230 °C |
| Source Temp. | 230 °C |
| Quad Temp. | 150 °C |
| Electron Energy | 69.9 eV |
| Solvent Delay Time | 1.00 min |
| Tune Type | BFB |
| Ionization Mode | EI |
| Scan Range | 35 – 250 amu |
| Scan Rate | 3.32 scans/sec |
Canister Cleaning/Blanks
Six SilcoCan air sampling canisters with Parker diaphragm valves and gauges were cleaned with a TO-Clean canister cleaning system. The canisters were cleaned per the recommended procedures outlined in section 8.4.1 of Method TO-15. Briefly, at 100 °C all canisters were evacuated down to 500 mTorr and held under vacuum for 60 minutes; the canisters were then filled with humidified (50% relative humidity [RH]) nitrogen to 30 psig. All RH values throughout this application note were verified by an EXTECH hygro-thermometer/datalogger (Model SDL500). This cycle was completed two additional times for a total of three cycles (Table III). All vacuums/pressures were controlled by the TO-Clean canister cleaning system’s internal gauge; however, vacuums/pressures were periodically verified using an Ashcroft digital test gauge (cat.# 24268).
Blank canisters were generated by pressurizing clean canisters with humidified (50% RH) nitrogen to 30 psig, storing them for 3 days at room temperature (this was done to simulate any sample handling/shipping times), and then analyzing them with a selected ion monitoring (SIM) method (this was done as to achieve better sensitivity compared to the scan method) for cleanliness. Nitrogen was humidified by sparging the nitrogen through a humidification chamber (cat.# 24282) filled with deionized (DI) water.
Table III: Canister cleaning procedures conducted at 100 °C with humidified nitrogen (50% RH).
| Cycle | Evacuation Vacuum (mTorr) | Pressurization Pressure (psig) |
|---|---|---|
| 1 | 500 (Hold for 60 min) | 30 |
| 2 | 500 (Hold for 60 min) | 30 |
| 3 | 500 (Hold for 60 min) | 30 |
| Final | 50 | 30 (Only for blanks) |
Calibration Curve
A five-point calibration curve was generated by analyzing a series of volumes of a 10.0 ppbv primary standard (Table IV). The default preconcentration volume was 400 mL. The 10.0 ppbv primary standard was generated by injecting 180 mL of TO-15 65 component mix (cat.# 34436) into an evacuated 6-liter SilcoCan air sampling canister and pressurizing the canister to 30 psig with 50% RH nitrogen. The standard was allowed to age for at least 24 hours, but was no older than 30 days at the time of use.
Table IV: Calibration curve.
| Standard (ppbv) | Injection Volume (mL) | Calibration Concentration (ppbv) |
|---|---|---|
| 10.0 | 400 | 10.0 |
| 10.0 | 300 | 7.50 |
| 10.0 | 200 | 5.00 |
| 10.0 | 100 | 2.50 |
| 10.0 | 40 | 1.00 |
Method Detection Limits
Method detection limits (MDLs) were determined as prescribed in the Code of Federal Regulations (40 CFR 136 Appendix B). Specifically, MDLs were determined from seven replicate measurements of a low-level standard containing each compound of interest at concentrations near (within a factor of five) the expected detection limits. MDLs were calculated as the standard deviation of the seven replicate measurements multiplied by 3.14 (i.e., the Student's t value for 99 percent confidence for seven values). MDLs were determined for the analytical system both in full scan and by selected ion monitoring (SIM) by the use of 0.20 ppbv and 75.0 pptv standards, respectively.
Precision
Replicate precision was determined from two canisters filled with a standard (5.00 ppbv and 50% RH) containing each compound of interest at concentrations near expected field sample concentrations. Precision for each analyte was calculated as the absolute value of the difference between the analyses of canisters divided by their average value and expressed as a percentage as follows:
Percent Difference (%) = |x1-x2|/μ x 100
x1 = First measurement value
x2 = Second measurement value
μ = Average of the two values
In addition, analytical precision determinations were made from seven replicate measurements of a standard (5.00 ppbv and 50% RH) containing each compound of interest at concentrations near expected field sample concentrations. Precision for each analyte was calculated as the standard deviation of the seven replicate measurements divided by the average value of the seven replicate measurements and expressed as a percentage as follows:
Relative Standard Deviation (RSD [%]) = σ/μ x 100
σ = The standard deviation of an array
μ = The average of an array
Audit Accuracy
Audit accuracy for each compound was determined from the analysis of an audit standard prepared at 10.0 ppbv and 50% RH. Audit accuracy was calculated as the difference between the nominal concentration of the audit standard and the measured value divided by the nominal concentration of the audit standard, expressed as a percentage as follows:
Audit Accuracy (%) = (Audit Value – Measured Value)/Audit Value x 100
Results and Discussion
The majority of laboratories performing Method TO-15 for VOC analysis of air samples use 60 m gas chromatography columns to separate the standard suite of 65 target analytes. More specifically, most laboratories are using a 60 m x 0.32 mm x 1.00 µm column and GC analysis times are on the order of 25 to 30 minutes. Total GC cycle times (i.e., including GC cool down) using a 60 m column are generally 30 to 35 minutes. While 60 m columns have been used historically, the long total cycle times are a significant disadvantage. The shorter analysis times and good peak resolution obtained using a 30 meter column are beneficial to labs interested in increasing sample throughput and overall productivity.
As shown in Figure 1, the 30 m x 0.32 mm x 1.00 µm Rxi-5Sil MS column (cat.# 13654) was more than adequate to separate the standard suite of 65 TO-15 VOCs. GC analysis times were just 16.5 minutes, approximately half the time of a typical analysis on a 60 m column. Some coelutions were observed, but these compounds coeluted identically on a 60 m column; further, since the compounds that coeluted are non-isobaric they can easily be distinguished with MS detection. In addition, much narrower peak widths and increased peaks heights were obtained, resulting in increased analytical sensitivity. With the use of Nutech’s 8900DS preconcentrator, total sample throughput time (i.e., including total GC cycle time [16.5 minute GC analysis + ~5 minute GC cool down] and sample preconcentration time [~15 minute for 400 mL]) was only ~22 minutes. Total sample throughput time was equivalent to total GC cycle time, due to the fact that while the GC analysis was being carried out on one sample, the next sample was preconcentrating. The use of a 30 m x 0.32 mm x 1.00 µm Rxi-5Sil MS column for Method TO-15 analysis increases sensitivity and decreases sample analysis times, thereby increasing sample throughput and reducing operating costs.
Figure 1: Analysis of 65 TO-15 compounds in 16.5 minutes on a 30 m Rxi-5Sil MS column.
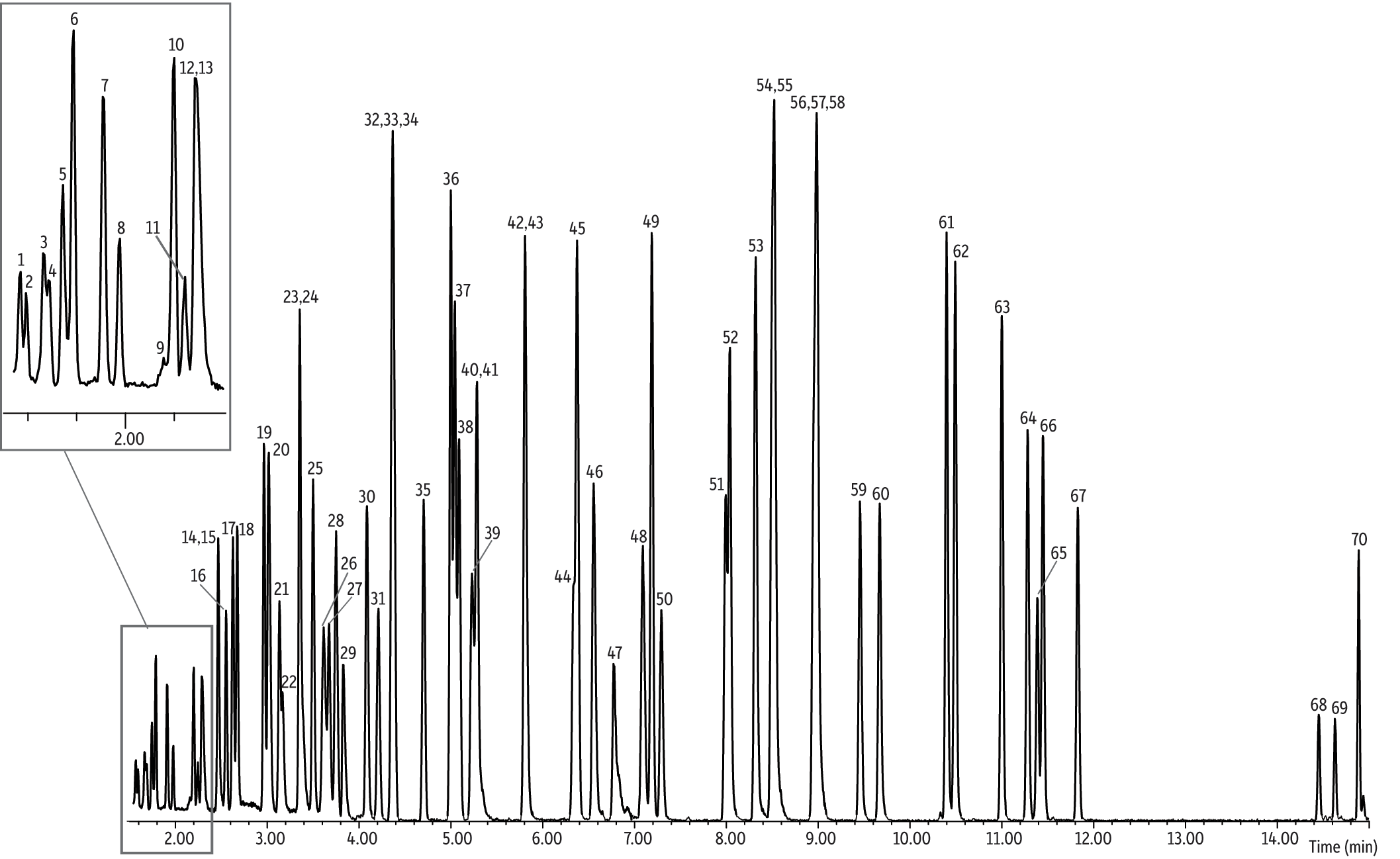
| Peaks | tR (min) | |
|---|---|---|
| 1. | Propylene | 1.57 |
| 2. | Dichlorodifluoromethane (Freon 12) | 1.60 |
| 3. | Chloromethane | 1.67 |
| 4. | 1,2-Dichlorotetrafluoroethane (Freon 114) | 1.68 |
| 5. | Vinyl chloride | 1.74 |
| 6. | 1,3-Butadiene | 1.79 |
| 7. | Bromomethane | 1.91 |
| 8. | Chloroethane | 1.98 |
| 9. | Ethanol | 2.16 |
| 10. | Trichlorofluoromethane (Freon 11) | 2.20 |
| 11. | Acrolein | 2.25 |
| 12. | Acetone | 2.29 |
| 13. | Acetonitrile (contaminant) | 2.29 |
| 14. | 1,1-Dichloroethene | 2.47 |
| 15. | Isopropyl alcohol | 2.49 |
| 16. | 1,1,2-Trichlorotrifluoroethane (Freon 113) | 2.55 |
| 17. | Methylene chloride | 2.63 |
| 18. | Carbon disulfide | 2.68 |
| 19. | trans-1,2-Dichloroethene | 2.97 |
| 20. | Methyl tert-butyl ether (MTBE) | 3.02 |
| 21. | 1,1-Dichloroethane | 3.13 |
| 22. | Vinyl acetate | 3.17 |
| 23. | 2-Butanone (MEK)* | 3.36 |
| Peaks | tR (min) | |
|---|---|---|
| 24. | Hexane* | 3.36 |
| 25. | cis-1,2-Dichloroethene | 3.50 |
| 26. | Ethyl acetate | 3.62 |
| 27. | Bromochloromethane (IS) | 3.67 |
| 28. | Chloroform | 3.75 |
| 29. | Tetrahydrofuran | 3.83 |
| 30. | 1,1,1-Trichloroethane | 4.09 |
| 31. | 1,2-Dichloroethane | 4.21 |
| 32. | Benzene | 4.36 |
| 33. | Carbon tetrachloride | 4.37 |
| 34. | Cyclohexane | 4.39 |
| 35. | 1,4-Difluorobenzene (IS) | 4.70 |
| 36. | Heptane | 5.00 |
| 37. | Trichloroethylene | 5.04 |
| 38. | 1,2-Dichloropropane | 5.09 |
| 39. | Methyl methacrylate | 5.23 |
| 40. | Bromodichloromethane | 5.28 |
| 41. | 1,4-Dioxane | 5.32 |
| 42. | 4-Methyl-2-pentanone (MIBK) | 5.81 |
| 43. | cis-1,3-Dichloropropene | 5.81 |
| 44. | trans-1,3-Dichloropropene | 6.33 |
| 45. | Toluene | 6.37 |
| 46. | 1,1,2-Trichloroethane | 6.55 |
| Peaks | tR (min) | |
|---|---|---|
| 47. | 2-Hexanone (MBK) | 6.77 |
| 48. | Dibromochloromethane | 7.09 |
| 49. | Tetrachloroethene | 7.19 |
| 50. | 1,2-Dibromoethane | 7.29 |
| 51. | Chlorobenzene-d5 (IS) | 7.99 |
| 52. | Chlorobenzene | 8.04 |
| 53. | Ethylbenzene | 8.32 |
| 54. | m-Xylene | 8.52 |
| 55. | p-Xylene | 8.52 |
| 56. | Styrene | 8.95 |
| 57. | o-Xylene | 8.98 |
| 58. | Bromoform | 9.00 |
| 59. | 1,1,2,2-Tetrachloroethane | 9.46 |
| 60. | 4-Bromofluorobenzene** | 9.67 |
| 61. | 4-Ethyltoluene | 10.40 |
| 62. | 1,3,5-Trimethylbenzene | 10.49 |
| 63. | 1,2,4-Trimethylbenzene | 11.00 |
| 64. | 1,3-Dichlorobenzene | 11.28 |
| 65. | Benzyl chloride | 11.39 |
| 66. | 1,4-Dichlorobenzene | 11.45 |
| 67. | 1,2-Dichlorobenzene | 11.83 |
| 68. | 1,2,4-Trichlorobenzene | 14.46 |
| 69. | Naphthalene | 14.63 |
| 70. | Hexachlorobutadiene | 14.89 |
| Column | Rxi-5Sil MS, 30 m, 0.32 mm ID, 1.00 µm (cat.# 13654) |
|---|---|
| Standard/Sample | TO-15 65 component mix (cat.# 34436) |
| TO-14A internal standard/tuning mix (cat.# 34408) | |
| Diluent: | Nitrogen |
| Conc.: | 10.0 ppbv 400 mL injection |
| Injection | Direct |
| Oven | |
| Oven Temp.: | 32 °C (hold 1 min) to 150 °C at 9 °C/min to 230 °C at 33 °C/min |
| Carrier Gas | He, constant flow |
| Flow Rate: | 1.5 mL/min |
| Linear Velocity: | 44 cm/sec @ 32 °C |
| Detector | MS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Mode: | Scan | ||||||||
| Scan Program: | |||||||||
| |||||||||
| Transfer Line Temp.: | 230 °C | ||||||||
| Analyzer Type: | Quadrupole | ||||||||
| Source Temp.: | 230 °C | ||||||||
| Quad Temp.: | 150 °C | ||||||||
| Electron Energy: | 69.9 eV | ||||||||
| Solvent Delay Time: | 1.0 min | ||||||||
| Tune Type: | BFB | ||||||||
| Ionization Mode: | EI | ||||||||
| Instrument | HP6890 GC & 5973 MSD | ||||||||
| Acknowledgement | Nutech | ||||||||
Table V: Results from blank, calibration, MDL, precision, and accuracy experiments demonstrate Method TO-15 criteria were met by the analytical system.
| Analyte | Average Blank Concentration (pptv)1 |
Calibration (%RSD)2 | Scan MDL (ppbv)3 | SIM MDL (pptv)4 | Replicate Precision (%Difference)5 |
Precision (%RSD)6 | Audit Accuracy (%)7 |
|---|---|---|---|---|---|---|---|
| Propylene | BDL | 8.51 | 0.10 | 66.9 | 1.69 | 9.08 | 12.8 |
| Dichlorodifluoromethane (Freon 12) | BDL | 6.22 | 0.05 | 33.4 | 1.79 | 7.82 | 1.74 |
| Chloromethane | BDL | 7.63 | 0.02 | 38.8 | 0.89 | 6.59 | 1.29 |
| 1,2-Dichlorotetrafluoroethane (Freon 114) | BDL | 18.9 | 0.08 | 65.3 | 3.33 | 7.71 | -1.99 |
| Vinyl chloride | BDL | 5.60 | 0.05 | 37.6 | 0.15 | 7.12 | -7.24 |
| 1,3-Butadiene | ND | 6.44 | 0.15 | 34.0 | 3.25 | 5.12 | -5.06 |
| Bromomethane | ND | 6.86 | 0.05 | 26.4 | 3.63 | 5.84 | -4.86 |
| Chloroethane | ND | 10.5 | 0.06 | 78.0 | 3.30 | 6.07 | -5.34 |
| Ethanol | 160 | 21.4 | 0.19 | 94.6 | 6.34 | 9.01 | -4.06 |
| Trichlorofluoromethane (Freon 11) |
BDL | 17.2 | 0.08 | 21.9 | 5.25 | 10.8 | -0.63 |
| Acrolein | BDL | 9.96 | 0.09 | 31.0 | 1.04 | 6.70 | -10.7 |
| Acetone | BDL | 10.8 | 0.14 | 45.1 | 6.60 | 5.55 | 1.20 |
| Isopropyl alcohol | BDL | 13.2 | 0.05 | 50.9 | 8.50 | 10.2 | 5.79 |
| 1,1-Dichloroethene | ND | 14.5 | 0.03 | 23.4 | 3.53 | 6.07 | -1.54 |
| 1,1,2-Trichlorotrifluoroethane (Freon 113) | BDL | 25.0 | 0.09 | 23.5 | 4.45 | 5.84 | 7.99 |
| Methylene chloride | BDL | 12.7 | 0.05 | 56.3 | 4.75 | 5.68 | 2.11 |
| Carbon disulfide | BDL | 7.12 | 0.03 | 38.0 | 5.14 | 7.61 | -1.89 |
| trans-1,2-Dichloroethene | ND | 8.14 | 0.05 | 39.9 | 4.89 | 6.46 | 0.37 |
| Methyl tert-butyl ether (MTBE) | ND | 5.17 | 0.03 | 42.8 | 3.41 | 6.53 | -2.74 |
| 1,1-Dichloroethane | ND | 18.4 | 0.03 | 25.2 | 4.23 | 6.36 | -5.87 |
| Vinyl acetate | ND | 2.94 | 0.05 | 33.0 | 1.22 | 7.06 | 1.94 |
| 2-Butanone (MEK) | ND | 7.47 | 0.06 | 39.9 | 6.07 | 7.34 | 0.89 |
| Hexane | BDL | 11.8 | 0.02 | 37.8 | 0.27 | 6.91 | -6.81 |
| cis-1,2-Dichloroethene | ND | 4.88 | 0.02 | 21.7 | 3.22 | 5.67 | -0.80 |
| Ethyl acetate | ND | 3.28 | 0.08 | 99.4 | 2.93 | 13.6 | -4.63 |
| Chloroform | ND | 11.6 | 0.02 | 11.9 | 4.47 | 6.64 | -1.51 |
| Tetrahydrofuran | ND | 7.97 | 0.08 | 41.6 | 0.12 | 9.72 | 6.01 |
| 1,1,1-Trichloroethane | BDL | 22.6 | 0.04 | 15.4 | 3.28 | 6.22 | -4.70 |
| 1,2-Dichloroethane | ND | 5.67 | 0.04 | 15.7 | 3.67 | 5.34 | 2.94 |
| Benzene | BDL | 8.92 | 0.02 | 61.2 | 9.55 | 6.60 | -1.17 |
| Carbon tetrachloride | BDL | 27.5 | 0.04 | 38.8 | 4.85 | 6.04 | 2.33 |
| Cyclohexane | ND | 29.8 | 0.05 | 40.7 | 4.00 | 5.61 | -0.16 |
| Heptane | ND | 3.71 | 0.04 | 28.0 | 13.55 | 5.41 | -2.46 |
| Trichloroethylene | BDL | 3.85 | 0.03 | 18.4 | 0.96 | 5.95 | -0.09 |
| 1,2-Dichloropropane | ND | 3.72 | 0.03 | 24.6 | 1.36 | 6.48 | 0.47 |
| Methyl methacrylate | ND | 18.6 | 0.14 | 20.7 | 3.53 | 7.75 | -1.63 |
| 1,4-Dioxane | ND | 11.5 | 0.08 | 19.6 | 0.13 | 7.10 | 0.90 |
| Bromodichloromethane | ND | 4.53 | 0.04 | 22.6 | 2.04 | 7.08 | 2.71 |
| 4-Methyl-2-pentanone (MIBK) | ND | 2.46 | 0.08 | 24.3 | 6.87 | 6.24 | 1.90 |
| cis-1,3-Dichloropropene | BDL | 4.05 | 0.04 | 8.30 | 0.80 | 7.59 | -0.86 |
| trans-1,3-Dichloropropene | ND | 2.44 | 0.05 | 20.4 | 8.30 | 5.86 | 0.79 |
| Toluene | BDL | 4.98 | 0.03 | 17.0 | 6.70 | 5.67 | -3.04 |
| 1,1,2-Trichloroethane | BDL | 4.30 | 0.05 | 14.3 | 0.58 | 5.73 | -1.64 |
| 2-Hexanone (MBK) | ND | 10.2 | 0.11 | 94.5 | 4.82 | 8.15 | 2.91 |
| Dibromochloromethane | BDL | 3.27 | 0.03 | 33.3 | 3.68 | 6.02 | 1.46 |
| Tetrachloroethene | BDL | 2.70 | 0.00 | 18.7 | 0.88 | 5.98 | 4.21 |
| 1,2-Dibromoethane | BDL | 2.28 | 0.04 | 17.6 | 7.85 | 6.63 | 1.77 |
| Chlorobenzene | ND | 8.27 | 0.05 | 17.4 | 2.93 | 4.91 | -3.37 |
| Ethylbenzene | BDL | 20.3 | 0.03 | 34.3 | 4.01 | 6.10 | -12.3 |
| m-Xylene | BDL | 6.00 | 0.04 | 12.1 | 5.51 | 6.70 | -14.3 |
| p-Xylene | BDL | 5.91 | 0.04 | 13.1 | 5.51 | 6.70 | -14.3 |
| Styrene | ND | 1.60 | 0.05 | 29.2 | 3.34 | 6.89 | -17.7 |
| o-Xylene | ND | 6.38 | 0.02 | 24.7 | 3.76 | 7.50 | -13.9 |
| Bromoform | BDL | 3.09 | 0.05 | 12.1 | 5.88 | 6.34 | -13.0 |
| 1,1,2,2-Tetrachloroethane | BDL | 5.87 | 0.06 | 20.4 | 8.30 | 7.99 | -9.79 |
| 4-Ethyltoluene | ND | 3.01 | 0.05 | 59.1 | 7.03 | 7.63 | -16.8 |
| 1,3,5-Trimethylbenzene | BDL | 4.13 | 0.05 | 49.5 | 5.98 | 6.43 | -17.1 |
| 1,2,4-Trimethylbenzene | ND | 1.86 | 0.07 | 68.2 | 5.09 | 4.92 | -14.1 |
| 1,3-Dichlorobenzene | ND | 5.18 | 0.07 | 33.7 | 5.75 | 7.07 | -11.8 |
| Benzyl chloride | ND | 23.2 | 0.05 | 44.2 | 4.58 | 7.42 | -13.5 |
| 1,4-Dichlorobenzene | BDL | 3.04 | 0.06 | 36.8 | 7.78 | 6.66 | -11.8 |
| 1,2-Dichlorobenzene | BDL | 6.26 | 0.07 | 36.4 | 6.92 | 7.72 | -11.9 |
| 1,2,4-Trichlorobenzene | ND | 15.9 | 0.24 | 39.0 | 7.39 | 6.42 | 11.0 |
| Naphthalene | ND | 17.7 | 0.15 | 70.3 | 3.37 | 6.82 | 15.1 |
| Hexachlorobutadiene | ND | 6.58 | 0.17 | 20.7 | 6.32 | 3.18 | 3.03 |
ND – Not detected
Results from blank, calibration, MDL, precision, and accuracy experiments are shown in Table V and discussed relative to specific method requirements below. Overall, excellent performance was obtained on the 30 m column in much shorter analysis times than are typically observed when using a 60 m column.
Canister cleaning/blank analyses demonstrate the TO-Clean canister cleaning system generated blank canister concentrations that met the Method TO-15 requirement (i.e., blank concentrations of less than 0.2 ppbv) for all 65 target analytes. The combination of heat and water vapor automated cleaning process and the hydrophobic/inert Siltek coating on the inside of the SilcoCan canisters helped to ensure the clean blanks. With the exception of ethanol, which averaged 160 pptv, all 65 components were either not detected or were below detection limits. Good result were obtained even for active compounds (e.g., acrolein), polar compounds (e.g., isopropyl alcohol, methyl ethyl ketone), and heavier semivolatile compounds (e.g., m- and p-xylene).
Calibration data demonstrate that the Nutech 8900DS preconcentrator, 30 m Rxi-5Sil MS column, and analytical system meet the Method TO-15 requirement (i.e., the %RSD for the RRF for each compound of interest was less than 30% with at most two exceptions up to a limit of 40%) for all 65 target analytes.
The %RSD for the average RRF was 9.31. These results also demonstrate that this system setup can provide linear concentrations of volumes spanning at least an order of magnitude (i.e., 40 to 400 mL).
Results from the method detection limits experiment show that the Method TO-15 MDL requirement (i.e., a method detection limit of ≤0.5 ppbv) was met for all 65 target analytes. The average scan and SIM MDLs were 0.06 ppbv and 35.9 pptv, respectively. It is important to note that the MDLs are strictly a statistical number (i.e., they do not always translate to real world concentrations with corresponding chromatographic peaks that are easily discernible from analytical noise); however, the system configuration used was able to adequately determine analytical peaks at 0.2 ppbv and 75.0 pptv for all 65 components in scan and SIM modes, respectively.
The replicate precision data demonstrate that the current method easily meets the Method TO-15 requirement (i.e., replicate precision within 25%) for all 65 target analytes. The average replicate precision was 4.29%. Although Method TO-15 (Section 11) stipulates that precision should be evaluated as the replicate precision of two canisters filled from the same air mass, this precision determination is not strictly evaluating the preconcentrator and analytical system. Rather, this precision includes any variability associated with sampling and/or storage. In addition, with only a two-point evaluation the precision could easily bias high or low. Therefore, in an attempt to provide a robust evaluation of the analytical precision (i.e., the preconcentrator and analytical system), a single standard was evaluated with seven replicate measurements and provided an average analytical precision of 6.86 %RSD. Similarly, results from the audit accuracy experiment demonstrate the system met the Method TO-15 requirement (i.e., audit accuracy within 30%) for all 65 target analytes. The average audit accuracy was -2.82%.
It is important to note that all results were obtained on a “close to real world” analytical system (i.e., although the instrument was tuned, the source had not just been freshly cleaned). In addition, the instrument used was a >10 year old 5973 with a standard turbo pump. Furthermore, except for instances in gross error (very infrequent), all chromatographic peaks were auto-integrated (i.e., the peaks were not manually adjusted).
This study clearly demonstrated that labs can shorten analysis times and, thus, increase productivity by moving from a 60 m column to a 30 m column and still meet Method TO-15 requirements. In addition, because this method is performance based, labs can also use different stationary phases if they wish to optimize separations. While excellent results can be obtained on a wide variety of 30 m columns—including Rxi-1ms, Rxi-5ms, Rxi-5Sil MS, and Rxi-624Sil MS columns—the Rtx-VMS column is recommended when polar or coeluting VOCs are included in the list of target analytes. The highly symmetrical peak shapes and lack of critical coelutions that can be achieved on this column will improve calibrations, MDLs, and replicate precisions. To see TO-15 VOCs analyzed on a 30 m Rtx-VMS column, reference article EVAR2388-UNV.
Conclusion
The following investigation was conducted to evaluate the efficacy of pairing Restek products with the Nutech 8900DS preconcentrator to meet the five most pertinent criteria of Method TO-15. Results demonstrate that the combination of the TO-Clean canister cleaning system, SilcoCan air sampling canisters, Nutech 8900DS preconcentrator, Agilent 6890/5973 GC-MS, and Rxi-5Sil MS column (30 m x 0.32 mm x 1.00 µm, cat.# 13654) easily meet all of the criteria for TO-15 VOC analysis. In addition, use of the 30 m x 0.32 mm x 1.00 µm column resulted in 16.5 min GC analysis times (~22 min total cycle times), which provide a significant time savings compared to a standard 60 m column. Obtaining method compliant results in less time presents an opportunity for increasing sample throughput for labs conducting TO-15 VOC analysis.

